physics of CYTOSKELETON & MORPHOGENESIS
CytoMorpho Lab
Laurent Blanchoin, Manuel Théry
Our work is dedicated to the investigation of the rules directing cytoskeleton self-organization. All cells have a cytoskeleton, a network of filaments that control cell shape and direct intra-cellular organization. As all components in cells, this network is dynamic, permanently assembling and disassembling, renewing its components and reconfiguring its architecture. This how cells sense and respond to external signals so that their internal organisation is adapted to the geometry of their environment.
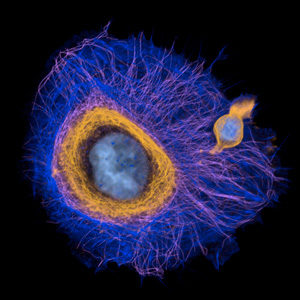
To reveal the rules directing cytoskeleton architectures we work on animal cells of various kinds as well as on reconstituted networks of filaments, that we assemble from purified proteins in cell-free systems. These two complementary approaches, complex and minimalist, physiological and fully controlled, allow us to reveal the core properties of living matter. In particular, we focus on the process of symmetry break and polarization of cytoskeleton networks. We aim at identifying the mechanisms by which cells orient in space and specify the axis that defines their body plan.
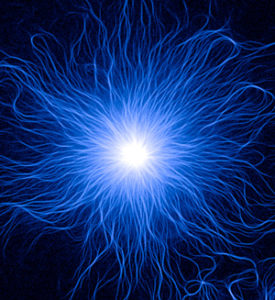


Our experimental strategy is to design microfabricated devices (surface micropatterning and microfluidic devices) in order to impose regular spatial boundaries to cells or networks of cytoskeleton filaments. Actin and microtubule network architectures appeared highly reproducible in those conditions, demonstrating that their assembly is driven by deterministic rules. We also use micro- and nano-devices devices to apply and measure mechanical forces. This is how we aim at describing the physics behind cytoskeleton architectures and hopefully formulate the corresponding rules in mathematical terms. By focusing on the contribution of force production in cytoskeleton networks and the role of symmetry elements we hope to unravel the basic mechanisms of cell polarisation.

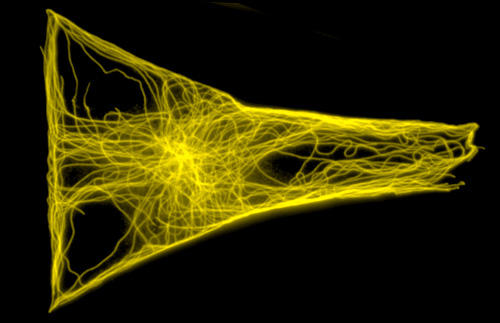
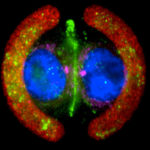
As we investigated cell architectures, we defined the geometrical laws regulating the orientation of the mitotic spindle, the positioning of centrosome, the clustering extra-centrosomes, the assembly of primary cilia, the velocity of cell motility, the magnitude and orientation of contractile forces as well as the positioning of inter-cellular junctions.
Looking in vitro at the self-organization of cytoskeleton filaments from purified components, we described the geometric rules that defines the alignment and contraction of actin filaments, the fracture and destruction of dense networks of actin filaments, the assembly of actin filaments at the centrosome as well as the self-repair of microtubules.


Cytoskeleton morphogenesis relies on spatial and temporal orchestration. The integration of local forces in the entire cellular network and the crosstalk between actin filaments and microtubules hold the key to a complete description of cell polarization. The characteristic times of actin and microtubule networks renewal and reorganization define how fast and how long cells can sense and remember the signals they have been submitted to.
Publications
Events
Job offer
OUR LAST PUBLISHED WORK
Happened recently in the lab
Join us







